Aluminum conducts current and is one of the best conductors in existence. It is used to make conductive busbars, cable lugs and sleeves, cable for overhead power lines, self-supporting insulated wire (self-supporting insulated wire) and wires of smaller cross-section (for domestic or industrial needs), coaxial television cable.
Types of aluminum electrical cable Source ru.prom.st
Advantages and disadvantages of copper wires
Copper is a ductile transition metal. It has a golden-pink color and is found in nature in the form of nuggets. It has been used by people since ancient times - an entire era was named in its honor.
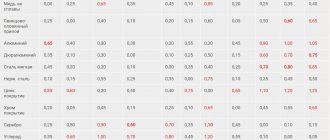
The table shows the electrical resistivity of steel and other metals
Today, copper wires are often used in electronic devices. Their advantages include:
- High electrical conductivity (the metal ranks second in this indicator, second only to silver). Compared to aluminum, copper is 1.7 times more efficient: with an equal cross-section, a copper cable passes more current.
- Welding, soldering and tinning can be carried out without the use of additional materials.
- The wires have good elasticity and flexibility, they can be rolled and bent without much harm.
Copper is only slightly inferior to silver.
However, until recently, copper wires were inferior to aluminum due to several disadvantages:
- High density: at different sizes, copper wire will weigh more than aluminum;
- Price: aluminum is several times cheaper;
- Copper oxidizes in open air: however, this does not affect its operation and is easily eliminated.
Other properties
Today, aluminum is produced almost twice as much as copper. And in comparison with all mined metals, it is second only to steel. This confirms that every year the electrical industry is increasing its use. This is explained by a number of reasons, which we will consider further.
Electrical performance of aluminum
According to the International Annealed Copper Standard (IACS), the latter is rated as 100% conductive. According to the above information, aluminum conducts electricity at only 61% equivalent to the generally accepted standard.
Thus, an equal percentage will only be achieved with larger cross sections. Since copper is significantly heavier than aluminum, such an “increased” conductor in mass will still be lighter than copper.
This fact has been proven through complex mathematical calculations, the result of which shows that 1 kg. aluminum provides equal conduction speed to that of 2 kg. copper Therefore, if certain technical conditions for the size of conductors do not require this, copper is replaced with aluminum.
Healthy! If for use in home wiring the weight of an electric wire does not have a special role, then when used on overhead power lines (overhead power lines), the weight of the current-carrying conductors has a significant effect. Therefore, take the one that is lighter, that is, aluminum.
Strength index
Provided the cross-section is the same, copper conductors are stronger than aluminum conductors. Although, this figure can be easily increased by alloying or thermomechanical treatment, or by increasing the cross-section.
The values given in the table show that aluminum conducts current, but is inferior to copper in terms of “break strength”. However, it is able to withstand its own weight and does not overload the overhead power line supports as much as copper.
In addition, extruding aluminum involves obtaining cross-sections of complex shapes, which cannot be obtained from steel. Based on such objective reasons, new elements can be designed in such a way that they will be the most effective in comparison with acceptable analogues from other materials.
Corrosion resistance
Aluminum does not require additional painting or zinc coating to protect against corrosion. The natural oxide coating protects the metal from subsequent contact with oxygen in the air and prevents further oxidation.
Interesting! If the protective oxide layer is mechanically damaged, it is instantly restored naturally
Life time
The duration of operation depends on a number of conditions. First of all, it is temperature and humidity. Although the official figures are 30 years for copper and 15 for aluminum, in practice cables last much longer. As an example, we can cite houses built by Stalin or Khrushchev. Some of them still have their original electrical wiring. However, official information is announced in precisely these terms.
Interesting! Sometimes the opinion is expressed that such electrical wiring in the house is dangerous and can lead to fire as a result of overheating of the contacts. But this can happen to any metal, and the reason lies not in its properties, but in a poor connection or overload of the line. Similar incidents often happen in Soviet-built houses. When designing apartments in the 70-80s. of the last century, no one imagined that in a few decades they would be “filled” with electrical appliances requiring a larger cross-section.
What is the resistance of copper and aluminum
Aluminum is a lightweight metal that can be easily machined and cast. It has high electrical conductivity: it ranks 4th after silver, copper and gold.
Important! Despite a number of advantages (low cost, light weight, ease of processing, and others), in the long term, aluminum wires are less profitable than copper wires.
In electrical engineering, 2 terms have meaning:
- Conductivity: Responsible for the transfer of current from one point to another. The higher the conductivity of a metal, the better it transmits electricity. At +20 degrees, the conductivity of copper is 59.5 million siemens per meter (S/m), aluminum - 38 million S/m. The conductivity of a copper cable is practically independent of temperature.
- Electrical resistance: the higher this concept, the worse the substance will pass current. The resistivity of copper is 0.01724-0.0180 μOhm/m, aluminum - 0.0262-0.0295.
In other words, copper has higher conductivity and lower resistance than aluminum.
What is the resistivity of steel
Steel is a metal alloy of iron with carbon and other elements. It contains at least 45% iron, the carbon content ranges from 0.02% to 2.14%. Depending on the exact composition, steel is used in construction, mechanical engineering and instrument making, as well as in many areas, for example, in transport, the national economy, and in the production of household appliances.
Steel wires have low conductivity
The conductivity of steel is only 7.7 million S/m, the resistivity is 0.13 μOhm/m, that is, it is quite high. Steel conducts electricity poorly and is not used in the production of cables. However, you can often find an outer galvanized steel braid that protects the wires from mechanical stretching. Such protection is needed if the cable runs under a road or on unstable ground, if there is a risk of sharply pulling the wire.
STRUCTURE
Cubic face-centered structure. 4 orange atoms
The crystal lattice of aluminum is a face-centered cube, which is stable at temperatures from 4°K to the melting point. There are no allotropic transformations in aluminum, i.e. its structure is permanent. The unit cell consists of four atoms with a size of 4.049596×10-10 m; at 25 °C, the atomic diameter (the shortest distance between atoms in the lattice) is 2.86 × 10-10 m, and the atomic volume is 9.999 × 10-6 m3/g-atom. Impurities in aluminum have little effect on the lattice parameter. Aluminum has great chemical activity; the energy of formation of its compounds with oxygen, sulfur and carbon is very high. In the voltage series, it is among the most electronegative elements, and its normal electrode potential is -1.67 V. Under normal conditions, interacting with atmospheric oxygen, aluminum is covered with a thin (2-10-5 cm) but durable film of aluminum oxide A1203, which protects against further oxidation, which determines its high corrosion resistance. However, if Hg, Na, Mg, Ca, Si, Cu and some other elements are present in aluminum or the environment, the strength of the oxide film and its protective properties are sharply reduced.
Comparison of conductivity of different types of steel
The characteristics of steel depend on its composition and temperature:
- For carbon alloys, the resistance is quite low: it is 0.13-0.2 μOhm/m. The higher the temperature, the greater the value;
- Low-alloy alloys have a higher resistance - 0.2-0.43 μOhm/m;
- High-alloy steels have high resistance - 0.3-0.86 μOhm/m;
- Due to the high chromium content, the resistance of chromium stainless alloys is 0.5-0.6 μOhm/m;
- Chromium-nickel austenitic steels are stainless and, thanks to nickel, have a high resistance of 0.7-0.9 μOhm/m.
Galvanized braiding is often made from steel.
Copper is in second place in terms of electrical conductivity: it perfectly passes electric current and is widely used in the manufacture of wires. Aluminum is also used no less often: it is weaker than copper, but cheaper and lighter.
Source
What to consider when choosing wiring
Copper and aluminum conduct electricity well. Most existing wiring is made from these metals. But there are differences between them. To decide what wiring is needed in your case, you need to consider the following factors:
- Copper wire can withstand more current, if we talk about an equal cross-section.
- Aluminum has a higher electrical resistivity. With the same transmitted powers, it heats up more than copper.
- Copper cables are more expensive. This metal is less common in nature.
- Aluminum is brittle. This causes difficulties during installation.
Aluminum cable APV 2x2.5
Aluminum wire, produced several decades ago, has qualitatively different mechanical properties. Even with the passage of time, it is softer and more comfortable. By this sign you can distinguish high-quality wiring.
Receipt
Aluminum is in first place among metals and in third place among all elements in terms of abundance in the earth's crust. Approximately 8% of the mass of the earth's crust is this metal. Aluminum is found in the tissues of animals and plants as a trace element. In nature, it is found bound in the form of rocks and minerals. The rocky shell of the earth, which is at the base of the continents, is formed precisely by aluminosilicates and silicates.
Aluminosilicates are minerals formed as a result of volcanic processes under appropriate high temperature conditions. During the destruction of aluminosilicates of primary origin (feldspars), various secondary rocks with a higher aluminum content (alunites, kaolins, bauxites, nephelines) were formed. Aluminum is included in secondary rocks in the form of hydroxides or hydrosilicates. However, not every aluminum-containing rock can be a raw material for alumina, a product from which aluminum is produced using the electrolysis method.
Aluminum is most often obtained from bauxite. Deposits of this mineral are common in countries of the tropical and subtropical zone. In Russia, nepheline ores are also used, deposits of which are located in the Kemerovo region and on the Kola Peninsula. When extracting aluminum from nephelines, potash, soda ash, cement and fertilizers are also produced along the way.
Bauxite contains 40-60% alumina. It also contains iron oxide, titanium dioxide, and silica. The Bayer process is used to isolate pure alumina. In an autoclave, the ore is heated with caustic soda, cooled, and the “red mud” (solid sediment) is separated from the liquid. Afterwards, aluminum hydroxide is precipitated from the resulting solution and calcined to obtain pure alumina. Alumina must meet high standards for purity and particle size.
Alumina (aluminum oxide) is extracted from the mined and enriched ore. The alumina is then converted into aluminum using electrolysis. The final stage is recovery by the Hall-Heroux process. The process is as follows: during the electrolysis of an alumina solution in molten cryolite, aluminum is released. The cathode is the bottom of the electrolysis bath, and the anode is carbon bars located in cryolite. Molten aluminum is deposited under a solution of cryolite with 3-5% alumina. The process temperature rises to 950°C, which is much higher than the melting point of aluminum itself (660°C). Deep purification of aluminum is carried out by zone melting or distillation through subfluoride.
Wire Specifications
The characteristics of the cables vary. Both metals have strengths and weaknesses. These parameters must be known for the correct selection, installation and maintenance of wiring in the apartment. To compare them, a number of criteria should be taken into account.
Electrical resistivity
This value shows the relationship between the conductor material and electrical resistance. This parameter determines what maximum current the cable can pass without overheating and melting the insulation.
| Metal | Electrical resistivity, Ohm*mm2/m |
| Copper | 0,017 |
| Aluminum | 0,028 |
From the table it follows that with equal lengths and cross-sections, the resistance of aluminum wires will be 1.67 higher. Hence the heating will be higher at equal currents.
Useful tips
In conclusion, here are a few tips that should be taken into account when organizing wiring:
- In case of independent design of wiring in a house or apartment, it is better to choose copper wires. With a smaller cross-section, they can withstand greater currents and are more resistant to frequent bending. An equally important point is volume. Copper wires are compact, which simplifies the process of creating grooves. For example, when connecting a receiver with a power of 7-8 kW, the aluminum wire should have a cross-section of about 8 mm. The cable has three cores plus a braid. As a result, the total diameter is about 1.5 centimeters. For comparison, copper can have a cross-section of 4 sq. mm, and the overall diameter is no more than a centimeter.
- When installing a socket, a three-core cable with a ground wire must be used. The distance of the socket from the floor is 30 cm. When organizing a lighting circuit, it is allowed to use cables with two cores (grounding is not needed here).
- It is forbidden to hang the entire load on one pair of wires (especially if they are aluminum). The best option is to divide the circuit into several lines. For example, a bathroom is powered through one machine, lighting through another, a kitchen through a third, and so on. The wire cross-section for the kitchen and bathroom should be 4 or 6 sq. mm, and for the lighting circuit - 1.5 or 2.5 mm.
The most difficult situation is in old apartments where aluminum wires are installed, which have outlived their useful life and require replacement. Wiring with a cross-section of 2.5 sq. mm can withstand a load of no more than 20 Amps, which is not enough for modern electrical receivers. In addition, the insulation of the wires loses its elasticity over time and gradually deteriorates. In such a situation, the only solution is to completely replace the wiring with copper wires.
For more information on why it is worth replacing aluminum wiring with copper in an old house, watch this video:
What kind of wiring is needed for an apartment?
In Soviet times, aluminum cables were used for wiring. The most powerful consumers of electricity were washing machines and refrigerators. They took a couple of hundred watts from the network. Aluminum coped with such low loads with a bang.
Now people use electric kettles (2 kW), vacuum cleaners (1-2 kW) and other powerful household appliances. Aluminum wires in such conditions overheat and burn out. Therefore, in a modern apartment you can only use copper wiring.
Additional Information. Regardless of whether aluminum or copper wiring is used, it is worth considering the insulation material. There must be compliance with fire safety requirements. Insulation is made from non-combustible materials. These standards are especially controlled in crowded places.
Advantages and disadvantages of copper wires
The use of copper requires PUE. Such wires are more suitable for transmitting electric current. They have the following advantages:
- high thermal and electrical conductivity;
- resistance to environmental influences;
- strength;
- ease of wiring.
According to the PUE, electrical wiring in residential premises should be done with copper cable
Copper and aluminum twist
It is strictly forbidden to twist aluminum cables with copper cables. These metals have different electrochemical properties. The resulting contact overheats, oxidizes and begins to burn. Hence all the ensuing consequences such as smoke and fire.
How to join copper and aluminum
For proper connection, you can use an intermediate conductor. Connect the copper and aluminum wire through an iron bolt with similar washers and nuts.
Bolted connection of copper and aluminum
Another common method is special Wago clamps with conductive lubricant. The connection will be significantly more expensive, but simpler, faster and more compact.
Features of connecting wires on the street
When installing a cable line along the street, all connection elements are exposed to external negative factors, such as snow, icing, rain, etc. Therefore, to perform such work, only a hermetically sealed structure that is resistant to ultraviolet rays and low temperatures is required. When making connections on a pole, roof or other open place, piercing clamps are most often used. You may be interested in learning in more detail how to connect SIP with copper cable on the street, because... in this case, the connection of aluminum and copper occurs in the open air.
In rooms, when laying a cable in a wall under plaster, the cable is laid in one piece in the groove, and any connection of even homogeneous metals is undesirable. All connections in a socket or junction box are made in any of the ways described above, suitable for each individual situation.