Modern electrical wiring in an apartment or house is carried out only with copper wires, as the PUE says. But in old houses, wiring was most often done with aluminum wire, and a situation arises in which it is necessary to connect 2 wires of different materials. And in this article you will learn how to connect copper and aluminum wire in different ways.
Methods for connecting copper and aluminum wires
Is it possible to twist copper wire with aluminum
Let's start with the fact that is it possible to connect aluminum wires with copper wires, and will such a connection not lead to a fire? The answer is yes, you can. But let's first get acquainted with these materials.
If you ask yourself which wiring is better, copper or aluminum, then the choice is, of course, copper. This comes out of the technical characteristics of copper; the cross-section of aluminum wire under the same conditions has to be taken larger. There are also disadvantages: copper is more expensive. It is easier to distinguish copper wire from aluminum by color; copper has a reddish tint, aluminum is gray and white.
Having looked at the electrical performance of metals, the question of which conducts current better disappears. Here's some information:
- Resistivity: copper – 0.017 Ohm mm²/m, aluminum – 0.028 Ohm mm²/m.
- Heat capacity: copper - 0.385 J/gK, aluminum - 0.9 J/gK.
- Elasticity of the material: copper – 0.8%, aluminum – 0.6%.
So why can’t you twist copper and aluminum wires, because twisting, especially with a small cross-section, is the cheapest option in terms of both money and time? The thing is that these materials, when connected, create a galvanic couple.
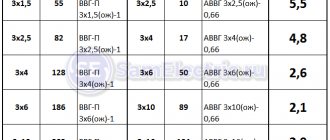
Galvanic pair - 2 metals of different kinds, the connection of which will lead to increased corrosion. Copper and aluminum are just such a galvanic pair. The electrochemical potentials of the two metals are too different, so rapid corrosion will increase the resistance at the junction and subsequent heating. More details about the compatibility of metals are indicated in GOST 9.005-72. Below is a table with some data on metals:
Galvanic compatibility of meltals
You can achieve high-quality contact between two conductors in different ways (soldering, using a simple terminal block, more expensive WAGO terminals, or an ordinary bolt and nut).
Materials and equipment used
Structure and properties of metals
The exception is cold galvanizing performed by Galvonol. This is a liquid suspension that is directly applied to the metal. It is unstable to some solvents and therefore requires a topcoat.
There are several groups of galvanic baths:
- Large ones. Designed for large-sized products.
- Average. They cannot accommodate a large product. At the same time, they remain the most in demand in conditions of medium-scale production.
- Small ones. They can only galvanize small parts.
Anode plates are placed in the bath. They are made from different materials. Their main task is to replenish the lost metal from the product during the galvanization process.
Important components are the type of electrolyte and current density. These parameters vary depending on the type of operation.
The compositions of cyanide baths for silver plating are presented in the table.
| Compound | Electrolyte number | |||
| 1 | 2 | 3 | 4 | |
| Cyanide silver | 2 | 6 | 30 | 100 |
| Sodium cyanide | 70 | 70 | — | — |
| Potassium cyanide | — | — | 70 | 100 |
| Sodium carbonate | 10 | 10 | — | — |
| Potash | — | — | 10 | 25 |
| Sodium hyposulfite | — | — | 0,4 | 0,5 |
| Ammonia aqueous, ml/l | — | — | 1-2 | 2 |
| Caustic potassium | — | — | — | 15 |
The current density influences the structure of the formed deposit. It is measured as the ratio of current per unit surface of the workpiece.
Connecting wires
Connecting aluminum and copper wires to each other requires technological solutions; simple twisting is not enough.
Methods for connecting conductors with different electrochemical potentials:
- By soldering. But not simple soldering.
- Using simple terminal blocks or expensive WAGO. There is no point in saving money here, and if the question is how to properly connect copper and aluminum wires, then it is better to take WAGO. The advantages of this manufacturer will be described below.
- Using a bolted connection, which has many advantages: low cost, simplicity and the ability to work with large cross-section wires.
- Crimping with sleeves. Requires specialized tools.
A little history
Luigi Galvani (1737-1798), whose name is given to the method of depositing parts of one metal onto the surface of another - electroplating - had nothing to do with the silvering of teaspoons and the galvanizing of aluminum buckets. He devoted himself entirely to theology, anatomy, physiology and physics, and was an outstanding physician of his time, who wanted to understand and explain the principles of “animal electricity”, when muscle contractions can be observed when current is passed through obsolete flesh. He was the first to describe the difference in potentials of different types of metals and electrolytes that arose during his experiments when they came into contact. He described and moved further in his electrophysiological research.
It occurred to the Russified German Moritz Hermann (1801-1874) to apply this difference for practical purposes. Having moved to Russia for permanent residence, Moritz changed his name to Boris, leaving behind the second of the ancient surnames that belonged to his family - Jacobi.
At the Imperial Academy of Sciences they laughed at Hermann a little, calling him “As if Boris” behind his back, but he also enjoyed enormous respect. It is he, a brilliant physicist and inventor, to whom Russia owes many brilliant inventions, who in 1840 wrote a work entitled: “A method for producing copper products using electricity using given samples from copper solutions or Electroplating for applied arts.”
It was with such a device that the history of the now widely used electrochemical process began, which has become a whole separate branch of modern industry.
WAGO
WAGO clamps for joining aluminum and copper are very popular, as they are very convenient to use:
- Click the pressure plates to the side.
- Insert wires into holes.
- Place the plates in their places and clamp them.
WAGO terminals for connecting copper wire to aluminum are an excellent solution
But now WAGO is casting doubt on its reputation. According to numerous reviews, the spring contact weakens, which leads to burning of the terminal block and its rapid replacement.
Conclusion
The zinc layer applied by hot-dip galvanizing can maintain its performance properties for up to 120 years when used under normal conditions. This is due to the thickness of the zinc layer, which is up to 200 microns. As a result, the metal acquires high protective properties and is resistant to mechanical stress. Moreover, the coating is capable of self-recovery when cracks form, which is due to the special composition of the zinc solution.
In turn, the thickness of the zinc layer during galvanization is no more than 15 microns. Therefore, the service life of products with such a coating thickness in aggressive conditions can last no more than 1 year. The advantages of this technique are affordable cost, evenness and uniformity of the coating.
Twisting wires
Twisting aluminum and copper wires was previously mentioned as a very unreliable connection method, but sometimes this is the only way to quickly restore power supply.
A couple of tips before twisting:
- Before twisting, the copper wire should be well tinned.
- The amount of twist must be at least 5 turns.
- After work, the joint must be protected with several layers of insulating tape or heat shrink tubing.
Soldering copper to the terminal block
You can solder copper and aluminum together. If everything is clear with copper, then for soldering aluminum you need a special flux. Some electricians simply solder copper wire to the terminal block.
Flux for aluminum
Terminal blocks
The list of electrician tools and consumables includes terminal blocks. Terminal blocks are copper or brass coated with a layer of nickel, designed for wires of a certain cross-section and covered with a layer of insulating plastic. The wires are secured with 2 small screws.
When connecting copper and aluminum terminal blocks, the locking screws must be properly tightened. If you tighten them, you can damage the aluminum conductors, which will not have a very good effect on the further operation of the electrical wiring. Therefore, it is necessary to find a middle ground: not tighten too tightly, but achieve high-quality contact.
Electroplating equipment
There is no need to “take away bread” from the owners of professional beauty salons. The corresponding techniques must be performed especially carefully so as not to cause harm to health. However, any ordinary person is able to prepare a high-quality set of equipment to solve technical problems.
The main component is a reliable and fairly powerful DC source. Adjustments in the desired range of voltage (1-12.5 volts) and current (up to 50-60 A) with a built-in indicator of the measuring device will be useful. The values of the required electrical parameters are selected after determining the operating settings of the technological operations.
A container with suitable dimensions is selected from a chemically neutral material. Heat-resistant plastic will do. However, it is better to use glass taking into account the following advantages:
- long-term preservation of consumer properties;
- strength, resistance to high temperatures;
- easy to clean.
Equipment set
As you can see in the photo, the electrodes can be fixed to the walls. The use of "crocodiles" speeds up the connection. To heat to the desired temperature, an electric stove with smooth power adjustment is useful. Scales are needed to accurately prepare the mixture.
Bolted connection
If you don’t have a terminal block, soldering iron or WAGO at hand, and the cross-section of the wires is large enough, then you can achieve high quality with an ordinary bolt.
To connect two wires you will need: a bolt, a nut, 3 washers. Sequencing:
- Make rings at the ends of the wires, the same diameter as the bolt. For convenience, it is better to use round nose pliers.
- Place the rings on the bolt in such an order that they are between the three washers.
- Tighten the nut and check the quality of the connection.
- Apply several layers of insulating tape.
Bolted connection of aluminum and copper
Copper corrosion in water
The corrosion rate of copper in water largely depends on the presence of oxide films on the surface.
In rapidly moving aqueous solutions and water, copper is subject to a type of destruction called impact corrosion. The rate of impact corrosion of copper is highly dependent on the amount of dissolved oxygen. If the water is highly aerated, impact corrosion of copper proceeds intensively, but if it is deoxygenated, the destruction is insignificant. Copper corrosion in aerated water increases with decreasing pH and increasing concentration of chlorine ions. The rate of copper corrosion in water depends on the climate zone. In the tropics, the rate of destruction is slightly higher.
A special feature of copper washed by sea water is that it is one of the few metals that are not susceptible to fouling by microorganisms. Copper ions are harmful to them.
Pipelines for supplying water to homes are often made from pure copper. They are reliable and last a very long time. In the presence of dissolved carbonic and other acids in water, copper gradually corrodes, and copper corrosion products stain plumbing equipment. If water passing through copper pipes comes into contact with iron, aluminum or galvanized steel, then the corrosion of these metals increases significantly. Copper ions are deposited on the surface of these metals, forming corrosive galvanic cells.
To eliminate the harmful effects of water from copper pipes on other metals, tinned copper is used. The inside of the copper pipeline is coated with tin. The tin coating must be non-porous to avoid the formation of a galvanic element (tin is a cathode in relation to copper).
Nut connection
"Nut" is another type of terminal block, most often used for branching large-gauge wires. It consists of 2 copper plates placed in a plastic case.
Copper and aluminum wire, as well as a branch wire, are placed between the plates. But you can simply use the “nut” as a connecting element. After laying the conductors, the plates are tightened with bolts. As insulation, a plastic case is placed over the entire structure, consisting of two halves, for fastening which standard screws are used.
Walnut connection is suitable for all types of street connections and branches
Brief history of the development and advantages of electroplating
Plating metals with copper at home
The date of development of the method of galvanic deposition (or rather, one of its varieties - galvanoplasty) is considered to be 1838, when it was invented by the famous scientist Boris Jacobi. After developing this technology, the scientist began to actively introduce it into various production processes, due to which mints and enterprises engaged in the production of printing equipment, as well as specialists in artistic crafts, began to use it.
In the Middle Ages, a galvanic bath was used to make copies of medals, which were dipped into a solution as a cathode (m)
Electroplating received its name not in honor of the inventor of this technology, Boris Jacobi, but in honor of the Italian scientist Luigi Galvani, who began to use the method of electrochemical processing of products almost simultaneously with Jacobi.
The most significant advantages of coating products with a layer of metal using galvanization include the following.
- Galvanic coatings can be applied without problems to parts with even very complex configurations.
- The coating formed using this technology is characterized by high density and uniform thickness.
- The coating applied by electroplating is characterized by excellent adhesion to the treated surface.
- The protective and decorative characteristics of galvanized coatings, if they are formed in strict accordance with technological requirements, are at the highest level.
- The thickness of the metal layer applied by electroplating can be easily adjusted.
Electroplating technology is so accessible that it allows you to independently create installations that are quite competitive with factory models
The term “plating” can be encountered not only in various industries and jewelry, but also in cosmetology. The process that is implied by this name of a cosmetic procedure can hardly be called galvanization in the full sense of the word, however, the term has taken root, and galvanic cleansing of facial skin is very popular in our time. When performing such cleansing, the skin is exposed to low-power currents, due to which the fats accumulated in its deep layers are liquefied and easily come out through the pores.
Crimping
For this method you will need special crimping pliers and sleeves. The principle of connecting wires with a sleeve is very simple: an aluminum wire is inserted into the sleeve on one side, a copper wire on the other, and the sleeve is crimped on both sides with pliers. There are sleeves for wires with a large cross-section - from 16 mm2 to 300 mm2, but in this case a special hydraulic press will be required. The only drawback of crimping is the high cost of the tool.
Special sleeve for connecting aluminum and copper
Galvanoplasty, galvanostegy, patination
Galvanoplasty is called copying technology. The essence of the processes does not differ from the above descriptions. However, adhesion is reduced to facilitate separation of the finished product from the workpiece.
Electroplating is an improvement in the mechanical parameters of the combined layer. Chrome, for example, prevents damage to steel products due to its high strength.
Patination is used to change the decorative properties of the surface. In particular, they create an artificially aged appearance.
Arrows mark areas created using the “rainbow” patination technology
Lubrication
To improve the quality of contact, you can use a special lubricant or paste. Usually this is quartz vaseline paste. It is usually used to improve the connection of aluminum wires.
But this paste can be used for all types of connections (threaded, using terminal blocks, crimping), especially if the connection takes place outdoors. Then the contact is affected by additional factors that significantly reduce the durability of the connection. Although the use of lubricant without insulation raises doubts.
Advantages and development history
Electrochemical activity series of metals
This technology was invented in 1838 by a scientist named Boris Jacobi. It was he who began the active introduction of galvanics into a variety of production processes. Soon, galvanic processing was successfully mastered by mints, artists, craftsmen, and industrial enterprises.
However, this technique received its name in honor of the Italian scientist Luigi Galvani. He began studying electrochemical processing technology almost simultaneously with Boris Jacobi.
The main advantages of galvanization include the following:
- Electroplated coatings are characterized by uniform thickness and the highest level of density.
- Electroplating can be easily applied even to structures with complex shapes.
- The coating resulting from galvanic treatment has good adhesion to many metals.
- The decorative and protective properties of galvanized parts are very high.
- The thickness of the galvanic coating is very easy to adjust.
By the way, the word “electroplating” is found not only in industrial fields and jewelry production, but also in cosmetology. This is the name of the process in which the skin is exposed to low-power currents to get rid of excess fat from the sebaceous glands.