Lead characteristics
The component is rarely found in its native form. There are more than 80 mineral rocks with the inclusion of the element, for example: cerussite, galena, anglesite.
Technological properties and characteristics of the metal
Lead has received the typical features and technological properties of its group and is characterized by increased heaviness and density with a traditional gray tint. When exposed to air, the metal loses its shine.
Despite its high density, the metal exhibits significant softness; at room temperature, you can leave a mark on it with your fingernail.
Lead density and mass
The mass of the element is 82, which is the reason for the large weight. The crystal lattice received a cubic shape. There is an atom in the corner of the molecule model and in the middle of all the faces.
Lead Characteristics
High mass places the substance on the list of heavy components. When heated, the density of the material decreases.
What is the melting temperature of lead?
Lead is not a refractory substance, which sets it apart from other dense elements. The substance can easily be deformed and rolled into foil.
The complete melting point of lead is 327.46 °C. To find out the exact melting temperature of lead, it is enough to use the formula F = 1.8 °C + 32. Thus, lead melting occurs at a temperature of 620.6 F.
The boiling point of the element occurs at – 1749 °C. To carry out the casting procedure, it is necessary to bring the component to 400–450 °C.
Lead casting
It is worth noting that at a temperature of -7.26 K, the component becomes superconductive. When melting, the component acquires fluid properties, viscosity increases and surface tension changes.
Mechanical properties
High ductility caused poor strength properties.
- Tensile strength is up to 13 MPa (iron has an indicator of 250 MPa).
- Brinell hardness is - 3.2 - 8 HB.
- Compression limit – up to 50 MPa.
- Elongation of the material is no more than 50-70%.
Thermal conductivity is two times less than that of Ferrum, 11 times lower than that of copper and amounts to 33.5 W/(m K). At room temperature, the heat capacity value is 0.12 kJ/(kg K).
Electrical conductivity exhibits poor electrical conductivity under normal conditions, with a resistivity of 0.22 ohm-sq. Mmm. For copper this figure reaches 0.017.
Corrosion resistance
In terms of its inert properties, lead approaches the category of noble metals. The high reaction threshold and the presence of an oxide film provide high resistance to rust. Sulfuric acid, as well as carbonic anhydride, hydrogen sulfide do not affect the element. In urban areas, corrosion levels can reach -0.00068 mm/year.
Lead is resistant to chromic, concentrated acetic, and phosphoric acid. The component is quickly destroyed in nitric and dilute acetic acid and concentrated sulfuric acid (90%).
Melting at home
Due to its low melting point, ancient people could melt cuprum over a fire and use the metal to make various products.
https://youtube.com/watch?v=LbSfTbupGMU
To melt copper at home you will need:
- charcoal;
- crucible and special tongs for it;
- muffle furnace;
- household vacuum cleaner;
- bugle;
- steel hook;
- melting mold.
The process proceeds in stages, the metal is placed in a crucible and then placed in a muffle furnace. The desired temperature is set, and the process is monitored through a glass window. During the process, an oxide film will appear in the container with Cu, which needs to be removed - open the window and move it to the side with a steel hook.
In the absence of a muffle furnace, copper can be melted using an autogenous furnace. Melting will occur if there is normal air access. A blowtorch melts brass and fusible bronze. The flame should cover the entire crucible.
The melting point of bronze and brass, like the melting point of copper and aluminum, is low.
Today, Cu is not used in its pure form in industrial settings. It contains many impurities: nickel, iron, arsenic, antimony, and other elements. The quality of the product is determined by the percentage of impurities in the alloy (no more than 1%). Important indicators are thermal and electrical conductivity. Due to its ductility, low melting temperature and flexibility, copper is widely used in many industries.
Ways to get rid of oxide
A protective film is formed on the surface of the material, consisting of a chemically stable compound of lead with atmospheric oxygen. Mechanical treatment allows you to temporarily remove plaque, which is restored in a few hours.
Interesting: Silumin - what kind of alloy is it and where is it used?
Lead oxide is not highly durable, but does not conduct electricity well, so battery terminals in cars are treated with a special lubricant that protects the metal from contact with atmospheric air.
In everyday life, to protect the surface cleaned with an abrasive tool, sunflower oil or a mixture of graphite lubricant and synthetic-based varnish are used. The parts are placed in a container with oil for 5-7 minutes, and then kept on a table previously covered with napkins or cloth. Graphite grease is used to protect lead or alloy parts used outdoors. To clean monolithic elements, it is allowed to use vinegar essence or nitric acid diluted with water.
For the manufacture of shot, an alloy based on lead and antimony is used (percentage content not exceeding 6%). To prevent film formation, it is recommended to store shot in sealed containers. Please note that contact with human skin accelerates the oxidation process. After washing the shot with acid solutions (a white precipitate may appear in the container, indicating the presence of antimony salts), it is necessary to treat the ammunition with an acidified solution of copper sulfate (a chemically inert film of copper is formed on the spherical surface).
Home and industrial methods
Without tin-lead solders (PLS), the existence of such an industry as radio engineering is impossible. Many industrial products contain POS coatings.
Tin-lead solders
The industry supplies the market with solder product:
- cast pigs;
- wire;
- foil tape;
- solder tubes with flux;
- powder or paste.
Alloys containing 90% tin and 10% lead are used for soldering products, which are then electroplated with gold or silver. The melting point of pure tin is 2310 C. Therefore, the solder will melt when heated to 2200 C.
Tubular solder with flux
Tin-lead POS with a predominance of tin in its composition (61%) has a lower melting point - 191%. POS 61 is used to cover contact groups in various devices; it is also used to process thin wire for windings of electric motor armatures and transformer coils.
Important! Taking into account the temperature at which tin melts, the % lead content in the alloy is regulated. This achieves a comfortable temperature regime, at which the tin-lead solder quickly turns into a liquid state
POS 30 melts at 256 degrees. The compounds are less durable than products with a higher tin content.
10 percent solder is far from the temperature threshold at which tin melts. Therefore, POS 10 is used as a durable material for tinning large metal surfaces.
Melt preparation and pouring
In industrial conditions, the melt is prepared in special crucibles, which are placed in electric furnaces (equipment equipped with electronic measuring equipment that maintains the desired melting mode).
In radio engineering production, special heating baths are used to prepare solder for printed circuit boards of radio circuits.
In workshops and at home, solder is melted with a soldering iron tip. To prepare a large volume of molten metal, it is placed in a copper vessel on an electric stove. The alloy in the form of scrap is loaded into the melting bath gradually, as the next layer of metal melts.
Fishing varnishes
Avid fishermen at home cast fishing sinkers and spoons by pouring molten tin into clay molds. The spoons are then coated with waterproof varnishes.
Fishing varnish
Melt preparation and melting point of lead
The melting point of lead allows the metal to be used for technical purposes for the manufacture of parts and the formation of alloys
Melting of the material can be carried out in artisanal conditions, while observing safety precautions
Preparing the melt
Melting of a material involves the process of transition from a solid to a liquid under the influence of a temperature gradient. This indicator depends on the characteristics of the components that form the alloy.
For example, the melting point of lead is 327 °C, and tin is 232 °C. For solder made from these materials, the liquid transition temperature is 183 °C.
Melting of materials occurs when heated. The rate of transition to a liquid state is called the melting point.
Lead is a plastic and viscous chemical element that is easy to process. It easily forms an oxide in air. A fresh cut of metal quickly becomes dull. The material is fragile and easily susceptible to mechanical stress.
The density of lead is 11.3 g/cm³. The specific heat of fusion of lead is 25 kJ/kg. The material is highly viscous and difficult to break into pieces. At the same time, it is very soft; it leaves a dent when pressed with a fingernail.
Metal can be easily cut with a knife.
Melting point is the temperature at which a metal turns into a liquid state.
The melting point of lead allows it to be heated in a ladle on a simple fireplace, followed by casting into molds.
The specific heat of lead at room temperature is 127.5 J/kg, and when the metal is heated to boiling, the figure increases.
Melting in artisanal conditions
- Lead, whose melting point is low, allows its use for casting a variety of crafts and sinkers for fishing at home. Forming a melt is not difficult, but basic safety and care must be observed.
- Metal melting should be carried out in a well-ventilated area. You can choose a hand burner as a heat source, and use a container made of a more durable and heat-resistant metal as a vessel.
- Having placed the material in the heating container, turn on the heat source at maximum power and direct the temperature flow closer to the material being melted. It will take some time to convert a significant amount of raw material into liquid.
- After turning off the burner, the molten material can be poured into the prepared casting mold. Wearing special gloves, carefully pick up the container with liquid, gently rotating it to prevent the formation of bubbles.
- It is necessary to pour metal into the mold at a distance so as not to burn exposed parts of the body with hot lead fumes. After pouring, leave the mold to cool to a safe temperature.
- Spilled melt can be easily mechanically removed from the surface using a screwdriver or chisel and used for the next melt.
- The material mixes well with other metals, which affects the composition and quality of the casting. When working, it is necessary to use special clothing and thick gloves to protect the skin of your hands from metal dust.
- Before pouring, you need to make sure that the mold is completely dry. In the presence of moisture, instantaneous evaporation may occur, which will result in the contact of the melt on the body.
Application of metal
Lead has been known to mankind for several millennia. Even in ancient Rome, it was used to make pipes for transporting water.
There are approximately 180 minerals in nature that contain chemical element No. 82. Lead deposits are often combined with ores of copper, bismuth, zinc, and silver.
Today, the use of metal in industrial production makes it possible to produce:
- battery plates;
- power cable sheaths;
- typographic fonts;
- alloys and solders;
- alloys for the manufacture of bearings;
- dyes;
- bullets and shot for hunting.
It is also used as a means of protection against radioactive radiation.
Lead is widely used in human life.
Until recently, the metal was used to increase the octane number of fuel and detect H2S, but gradually this method began to be abandoned.
Lead is a toxic chemical element. Poisoning by metal and its compounds is possible during the development of ore deposits, smelting and use in production.
Household poisoning occurs due to long-term storage of food in packages or dishes coated with glaze containing lead.
Application areas of lead alloys
Lead has been known to mankind for thousands of years. In ancient Rome it was used to create water pipes. It is used in various industries. Lead is made from:
- Protective sheath for power cables.
- Bearing connections.
- Dyes.
- Weights for fishing.
- Fonts for typography.
- Shot used for shooting from hunting rifles.
- Protective elements for objects contaminated by radiation.
- Alloys for soldering.
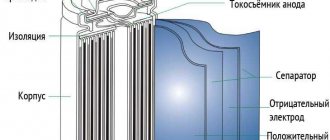
- Plates that are installed in batteries.
In the roofing industry, this material is often used to make soundproofing coatings.
Temperature - Melting - Lead
Fusibility is the ability of metals to change from a solid to a liquid state at a certain temperature. This temperature is called the melting point. Different metals have different, but specific (for a given metal) melting point. Alloys do not have a constant and definite melting point; it depends on the quantity and properties of the elements included in the alloy. So, for example, if the melting point of lead is 327 C, and tin is 232 C, then an alloy of tin (62%) with lead (38%), called solder, has a melting point of 183 C.
Fusibility is the ability of metals to change from a solid to a liquid state at a certain temperature. This temperature is called the melting point. Different metals have different, but specific (for a given metal) melting point. Alloys do not have a constant and definite melting point; it depends on the quantity and properties of the elements included in the alloy. So, for example, if the melting point of lead is 327 C, and tin is 232 C, then an alloy of tin in the amount of 62% with lead in the amount of 38% (the so-called solder) has a melting point of 183 C.
Fusibility is the ability of metals to change from a solid to a liquid state at a certain temperature. This temperature is called the melting point. Different metals have different, but specific (for a given metal) melting point. Alloys do not have a constant and definite melting point; it depends on the quantity and properties of the elements included in the alloy. So, for example, if the melting point of lead is 327 C, and tin is 232 C, then an alloy of tin (62%) with lead (38%), called solder, has a melting point of 183 C.
Crucible furnaces usually do not have a thermocouple or thermometer to measure temperature. The approximate temperature in the furnace is judged by the color of the heated ceramic muffle: dark red heat - 700 C, bright red - 950 C, yellow - 1100 C
After 10–12 minutes, the crucible is carefully removed, grasping it with crucible tongs. It must be emphasized that it is advisable to perform this operation without delay, without allowing the crucible to cool. When cooling, the metal can sometimes fuse to the crucible and is then difficult to remove without breaking the crucible.
For reduction, oxides of low-melting metals are chosen (melting point of lead 327 C, tin 232 C) to obtain molten metal in laboratory conditions
When cooled, the metal can sometimes fuse to the crucible and is then difficult to remove without breaking the crucible. For reduction, oxides of low-melting metals are chosen (melting point of lead 327 C, tin 232 C) in order to obtain molten metal in laboratory conditions.
Lead is plastic and tough, easy to process. A fresh cut of lead in air quickly becomes dull, as the lead is oxidized by oxygen in the air. Due to its high viscosity, lead is difficult to break. The mechanical strength of lead is very low. It is so soft that it can be drawn with a fingernail, easily cut with a knife, easily bent and torn even with relatively little effort. The thermal conductivity of lead between 0 and 50 is equal to 30 kcal/hour C, the heat capacity between O and 100 is equal to 0 031 kcal. C, linear expansion coefficient is 0 0000276 – 0 0000293, sp. The melting point of lead is 327 5, so it can very easily be melted in a ladle on a simple hearth and cast into any shape. Ease of processing is the most valuable property of lead compared to other materials.
This metal melts at a low temperature, which can be achieved using an ordinary gas burner or electric stove.
Properties of tin, its melting point
In nature, a rare metal can be found in two forms - in rocks and minerals. Most often, the element is found in the form of tin stone - an oxide compound. Previously, it was smelted from ore that was found in the upper layers of the earth's crust. Nowadays, such minerals have practically disappeared, so the process of mining tin has become much more difficult.
- Until the moment when the metal enters the smelting department, ore and placers that contain tin undergo an enrichment process. After this, the concentrate is sent to kilns and only then melted.
- The rare element has a low melting point; the melting process begins at +231.9°C; at a temperature of +231.0°C the metal remains solid. Even when cooled, it bends easily, and when heated it becomes pliable like plasticine. The boiling process of tin begins when the temperature is many times higher than the melting point - 2630 ° C.
- The element comes in white and gray colors; it acquires a darker color when it turns into a powder state; in powder, the density of the element is much lower than when it is in a solid state.
During the smelting process, slags, fluxes, and additives are used in order to obtain the desired grade and quality of metal. Its low melting point made it a strategically important metal. It can easily participate in the formation of alloys with other materials due to its low melting point. Ultimately, alloys are easily processed, then they participate in the connection of structural units and parts with a good sealed seam.
The effect of lead on the human body and symptoms of poisoning
Any lead compounds are very poisonous. The metal enters the body along with food or inhaled air and is carried by the blood. Moreover, inhaling lead vapors and dust is much more dangerous than its presence in food. Lead tends to accumulate in bones, partially replacing calcium in this case. When the concentration of lead in the body increases, anemia develops, the brain is affected, which leads to a decrease in intelligence, and in children it can cause irreversible developmental delays. It is enough to dissolve one milligram of lead in a liter of water and it will become not only unsuitable, but also dangerous for drinking. Such a low amount of lead also poses a certain danger; neither the color nor the taste of the water changes. Main symptoms of lead poisoning:
- gray border on the gums,
- lethargy,
- apathy,
- memory loss,
- dementia,
- vision problems,
- early aging.
Safety precautions
Molten lead can cause significant injuries and burns. The drop will instantly burn through the clothing and land on the exposed skin. The liquid form of lead can leak onto flammable objects and cause a fire in the room. If water penetrates into the liquid melt, a sharp flash occurs with the spread of small splashes of metal over the entire area. Such inclusions can get on the skin and eyes, which is painful and dangerous for human organs. Thus, while working, it is imperative to use clothing that covers the entire body, including sleeves, and wear a hat. The fabric must be highly fire-resistant and heat-resistant. You must wear a mask and safety glasses on your face.
The room in which smelting is carried out must be well ventilated, due to the toxicity of lead evaporation. If a mask is not available, use a cotton-gauze bandage. If lead enters the body, it can provoke and aggravate a number of painful processes, accumulating in organs, the element causes acute poisoning.
APPLICATION
Lead nitrate is used to produce powerful mixed explosives.
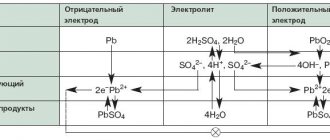
Lead azide is used as the most widely used detonator (initiating explosive). Lead perchlorate is used to prepare a heavy liquid (density 2.6 g/cm³) used in flotation beneficiation of ores, and it is sometimes used in high-power mixed explosives as an oxidizing agent. Lead fluoride alone, as well as together with bismuth, copper, and silver fluoride, is used as a cathode material in chemical current sources. Lead bismuthate, lead sulfide PbS, lead iodide are used as cathode material in lithium batteries. Lead chloride PbCl2 as a cathode material in backup current sources. Lead telluride PbTe is widely used as a thermoelectric material (thermo-emf 350 µV/K), the most widely used material in the production of thermoelectric generators and thermoelectric refrigerators. Lead dioxide PbO2 is widely used not only in lead batteries, but also on its basis many reserve chemical current sources are produced, for example, lead-chlorine cell, lead-fluorescent cell and others.
Lead white, basic carbonate Pb(OH)2•PbCO3, dense white powder, is obtained from lead in air under the influence of carbon dioxide and acetic acid. The use of white lead as a coloring pigment is no longer as common as it once was due to its decomposition by hydrogen sulfide H2S. Lead white is also used for the production of putty, in the technology of cement and lead carbonate paper.
Lead arsenate and arsenite are used in insecticide technology to kill agricultural pests (gypsy moth and cotton boll weevil).
Lead borate Pb(BO2)2•H2O, an insoluble white powder, is used to dry paintings and varnishes, and, along with other metals, as coatings on glass and porcelain.
Lead chloride PbCl2, a white crystalline powder, is soluble in hot water, solutions of other chlorides and especially ammonium chloride NH4Cl. It is used to prepare ointments for treating tumors.
Lead chromate PbCrO4 is known as chrome yellow dye and is an important pigment for making paints, for dyeing porcelain and fabrics. In industry, chromate is used mainly in the production of yellow pigments.
Lead nitrate Pb(NO3)2 is a white crystalline substance, highly soluble in water. This is a binder of limited use. In industry, it is used in matchmaking, textile dyeing and printing, antler dyeing and engraving.
Since lead absorbs γ radiation well, it is used for radiation protection in X-ray facilities and in nuclear reactors. In addition, lead is considered as a coolant in projects of advanced fast neutron nuclear reactors.
Lead alloys are widely used. Pewter (tin-lead alloy), containing 85–90% Sn and 15–10% Pb, is moldable, inexpensive, and used in the manufacture of household utensils. Solder containing 67% Pb and 33% Sn is used in electrical engineering. Alloys of lead and antimony are used in the production of bullets and typographic fonts, and alloys of lead, antimony and tin are used for figured casting and bearings. Lead-antimony alloys are commonly used for cable sheaths and electric battery plates. There was a time when cable sheaths used a significant portion of the world's lead production, due to the good moisture-proof properties of such products. However, lead was subsequently largely replaced from this area by aluminum and polymers. Thus, in Western countries, the use of lead on cable sheaths fell from 342 thousand tons in 1976 to 51 thousand tons in 2002. Lead compounds are used in the production of dyes, paints, insecticides, glass products and as an additive to gasoline in the form of tetraethyl lead (C2H5)4Pb (a moderately volatile liquid, the vapors of which in small concentrations have a sweetish fruity odor, in large concentrations - an unpleasant odor; Tm = 130 ° C, Bp = +80 °C/13 mm Hg; density 1.650 g/cm³; nD2v = 1.5198; insoluble in water, miscible with organic solvents; highly toxic, easily penetrates skin; MPC = 0.005 mg/ m³; LD50 = 12.7 mg/kg (rat, oral)) to increase octane number.
Used to protect patients from radiation from X-ray machines.
Lead - Pb
| Molecular weight | 207.20 g/mol |
| origin of name | from Latin plumbum |
| IMA status | valid, first described before 1959 (before IMA) |
general information
Lead-free solder, a type of soldering compound, is considered the most environmentally friendly option when joining metals. It contains no dangerous compounds or heavy metals. Since 2006, lead-free solders have been a mandatory requirement for electronics manufacturing in the EU and US. Compositions containing cadmium, mercury, and 6-valent chromium are not considered lead-free.
Melting point is one of the main differences between such compositions. In the case of lead-containing solders, the optimal temperature range for heating them is from +180 to +230 degrees Celsius. This allows you to use soldering irons without strict control over the thermal characteristics of the device. The compositions of the lead-free group are not so homogeneous. Some solders have a melting point below +180 degrees, but the bulk requires heating to +200... 250 degrees Celsius.
Rare earth and noble metals are used as lead replacement substances. Gold, silver, copper, and less commonly bismuth and indium are used. Copper-zinc compounds are also lead-free, but they have a higher melting point - over +300 degrees, and are not relevant for household use.
Due to their poorer wettability, lead-free solders require more careful application and flux selection. The seam created with their help also withstands less mechanical loads than when soldering with tin-lead compounds.
Which metal has the highest melting point
Tungsten is the most refractory metal, 3422 °C (6170 °F).
A hard, refractory, fairly heavy material of light gray color that has a metallic sheen. It is difficult to machine. At room temperature it is quite fragile and breaks. The fragility of the metal is associated with contamination with carbon and oxygen impurities.
Tungsten can be obtained from its ore, through complex chemical processing, as a powder. Using pressing and sintering, it is used to create parts of regular shape and bars.
Tungsten is an extremely resistant element to any temperature influences. For this reason, tungsten could not be softened for more than a hundred years. There was no furnace that could heat up to several thousand degrees Celsius. Scientists have been able to prove that this is the most refractory metal. Although there is an opinion that seaborgium, according to some theoretical data, has greater refractoriness, this is only an assumption, since it is a radioactive element and has a short lifespan.
Preparing to smelt lead
First you need to find a container. It would be great if the handle of the vessel was made of some heat-resistant material. For this purpose, you can use an old coffee pot or kettle.
The material can also be melted in an outdated container made of cast iron, using a deep and long spoon for pouring.
If there is no suitable container nearby, then you can use an ordinary tin can. However, here you should use pliers, which will be used to remove the hot pan from the flame and pour the material into the mold.
Do not forget that you need to be extremely careful while working. To simplify the procedure, you can make a small groove on one side of the jar. In this case, the red-hot metal will pour out in a thin stream clearly into the required place.
The material, cleared of impurities, can be crushed so that it melts as quickly as possible. The container must be securely placed over the burner and heated properly. This must be done in order to rid the surface of excess impurities and moisture.
Melting procedure
There is no need to try to melt all the prepared lead at once, because only the very bottom layer will interact with the hot surface of the container.
Melt two or three pieces first to form a puddle, then gradually add new material. This will give you the opportunity to make your work area more voluminous.
After melting, a layer of debris, impurities and slag must be removed from the surface of the metal. Pouring must be done into a heated mold. Lead is also characterized by rapid solidification. The material quickly loses its fluidity, becomes thicker, and therefore cannot completely fill the mold.
Read also: Lubricant for bolts with molybdenum disulfide
Security measures
When working with molten metal, it is imperative to protect the skin from thermal burns. During work, be sure to wear fireproof gloves and cloth or linen clothing that can withstand the ingress of molten substances.
In addition, it is necessary to wear a respirator or gauze bandage to protect the respiratory system - lead is a toxic substance.
So, by following these tips and using the information about the melting process, it won't be much of a problem to cast a fishing sinker or melt down the remains of an old battery into one ingot.